Sometimes substances that have long been used in industry or medicine are poorly explored, and scientists still get lost in their structures, features, and even generation mechanisms. Thus, chemists have known a substance called melamine cyanurate for a long time. These are crystals formed from a mixture of melamine and cyanuric acid.
Melamine cyanurate seems to be a well-studied substance. It is widely used to produce paint and varnish materials, impart fire-resistant properties to some plastics, and so on. Yet scientists still debate over the formation of this material. They are still not fully aware of what exactly happens to the molecules of cyanuric acid and melamine when under certain conditions they are placed in one container where nucleation begins.
The birth of crystals
A group of scientists, which included specialists from ITMO University and Moscow Institute of Physics and Technology, presented a study revealing molecular-level insights into melamine cyanurate self-assembly.
“This project is devoted to molecular modeling,” says Alexandra Timralieva, a co-author of the study and curator of the educational programs at ITMO’s Infochemistry Research Center. “We examined the nucleation process of the supramolecular complex of melamine cyanurate to pinpoint the specific features of such formations. Our research team decided to explore the structuring process from nucleation to crystal formation. It was revealed that variations of component concentration ratio can be used to influence the nucleation mechanism and thus the forming crystal. This will provide a better understanding of the peculiarities of its use for the delivery of biomolecules.”

While the main calculations were carried out by scientists in Moscow, the experiments were conducted at ITMO’s laboratories. The scientists investigated how changes in the concentration of one of the two components can affect the formation of melamine cyanurate.
Prospects
Although this study is fundamental and seemingly not practice-oriented, it may have significant implications for applied supramolecular chemistry – first and foremost, for the experiments on the introduction of drugs into supramolecular complexes for their targeted delivery.
Melamine cyanurate is useful in exploring the mechanism of drug introduction into crystals with a similar internal organization.
“We introduce active molecules into structures resembling melamine cyanurate to understand the nucleation process of supramolecular structures. This will help us make the process more meaningful,” explains Alexandra Timralieva. “Now, we plan to run model tests using numerous organic molecules such as tetracycline antibiotics.”
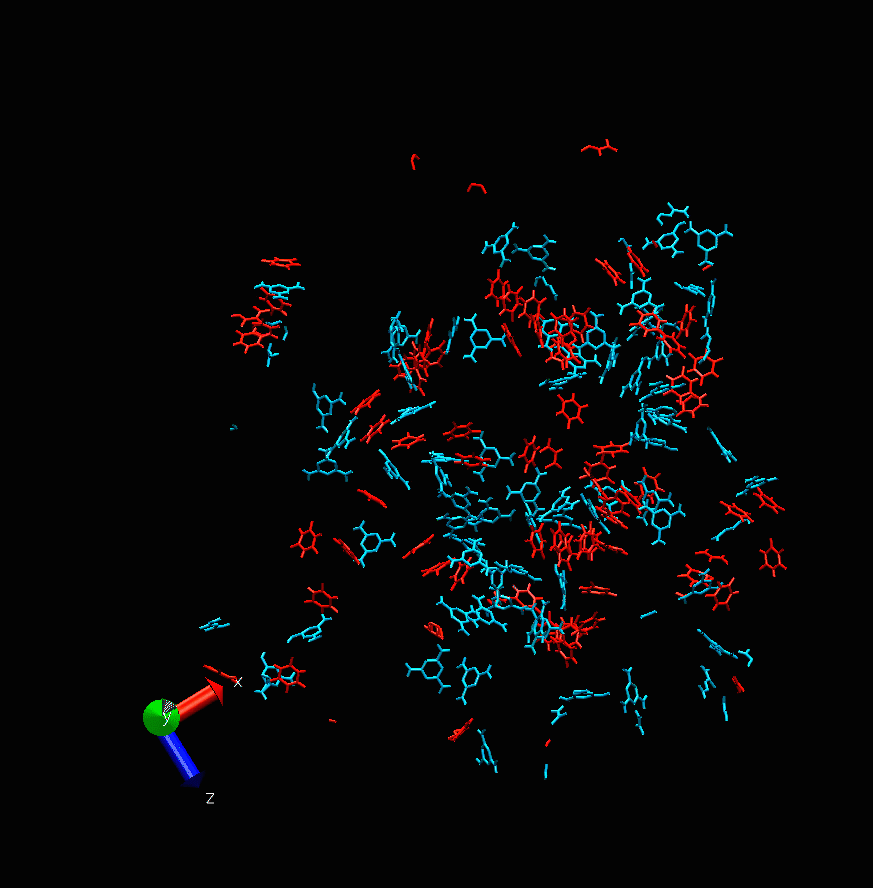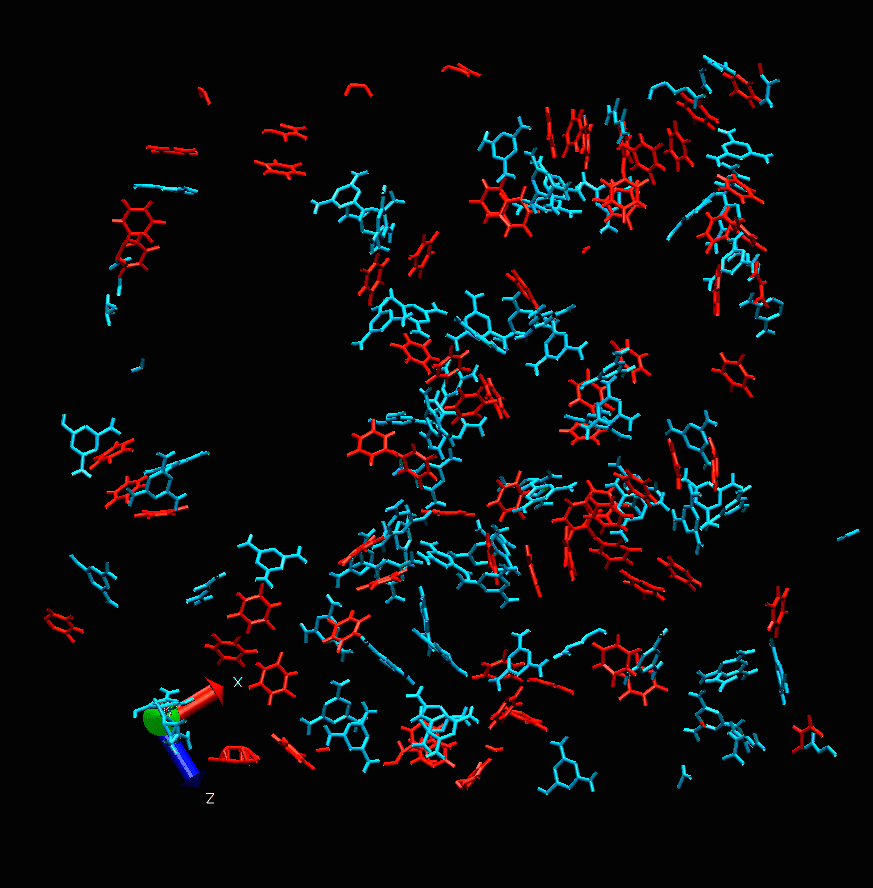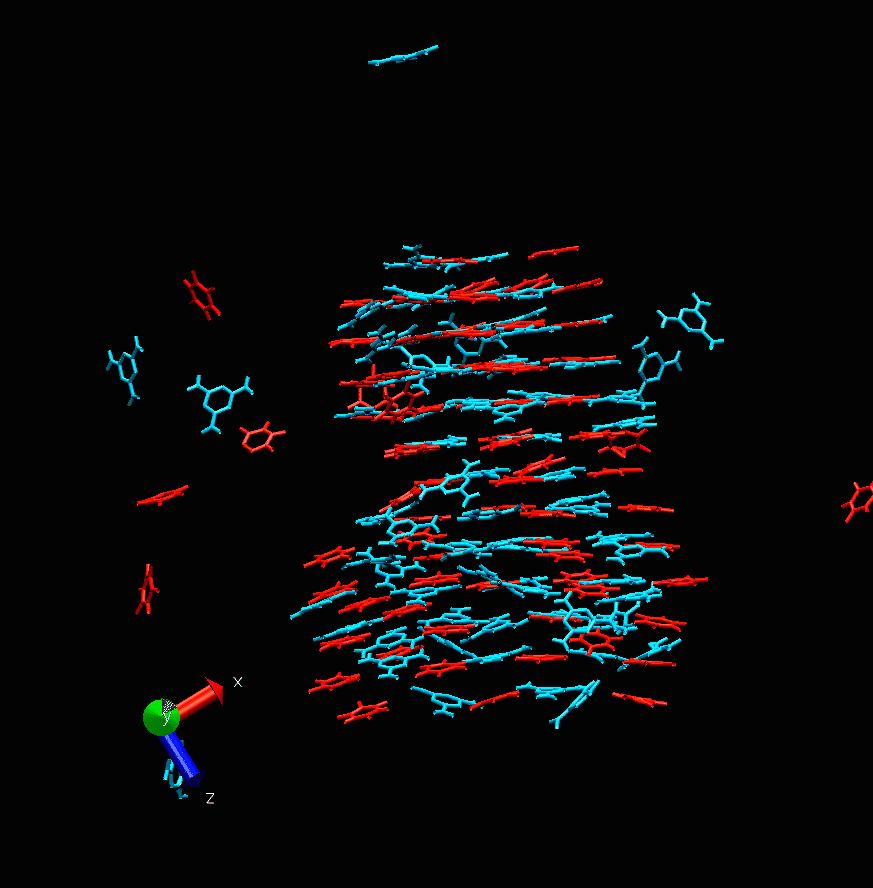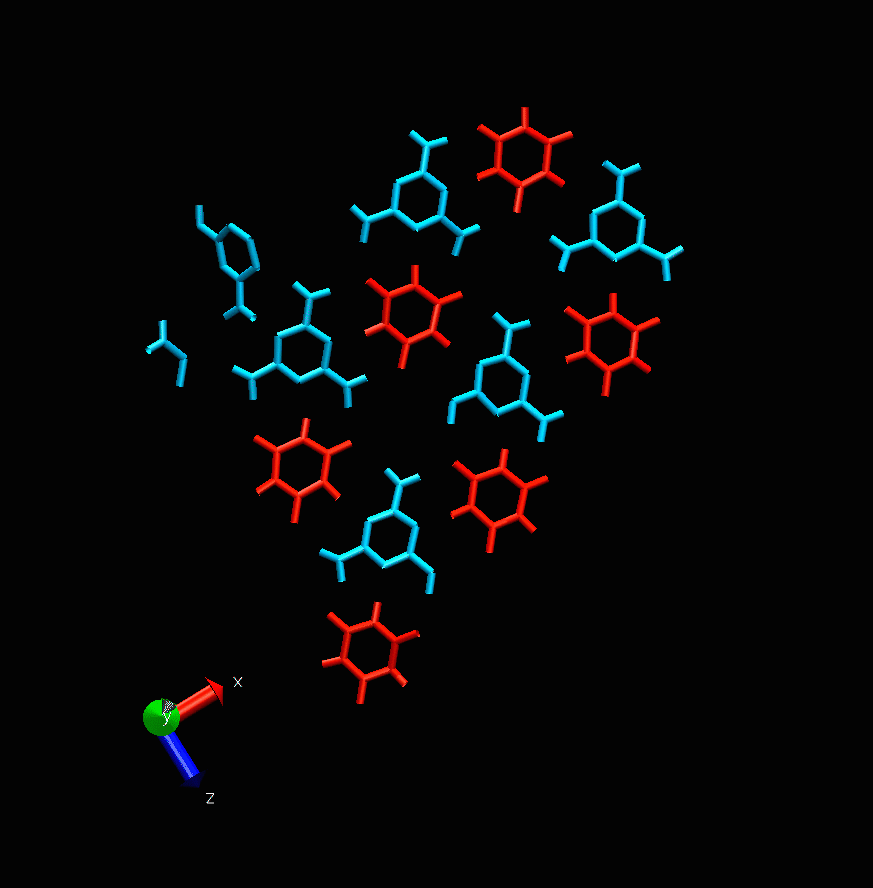
Melamine cyanurate nucleation. Photo courtesy of the scientists
This discovery can also be used to create an artificial DNA molecule, and such experiments are already underway. In 2008, American scientists announced the creation of the first artificial DNA. However, it is still under development, and the success of lab work will depend on the amounts of theoretical data in the hands of researchers.
“The formation of various supramolecular structures, including melamine cyanurate is very similar to that of DNA,” concludes Alexandra Timralieva. “If we figure out how to control the nucleation of such structures, then we will be able to move on to the chemistry of the origin of life. The chemical foundations of biological and supramolecular processes are not identical yet quite similar.”
The research is published in Crystal Growth & Design.
Reference: Nikita Orekhov, Nikolay Kondratyuk, Mikhail Logunov, Alexandra Timralieva, Vladimir Shilovskikh, and Ekaterina V. Skorb. Insights into the Early Stages of Melamine Cyanurate Nucleation from Aqueous Solution. Crystal Growth & Design, 2021/10.1021/acs.cgd.0c01285






