You have recently published an article. What were its main ideas?
The article covers immune response regulation in viral pneumonia. We were mainly led to analyze it closely by the current deteriorating situation with COVID-19 that is only made worse by other respiratory infections, seasonal flu, etc. Thus, we need a deeper level of understanding of the mechanisms behind our immune response in these kinds of illnesses.
How is viral pneumonia different from other types of pneumonia?
There are two main types of pneumonia: viral and bacterial. The former poses a greater risk because it results in strong cytokine storms with subsequent damage to lung tissue. Moreover, viral pneumonia can often be aggravated with a bacterial infection.
What is a cytokine storm?
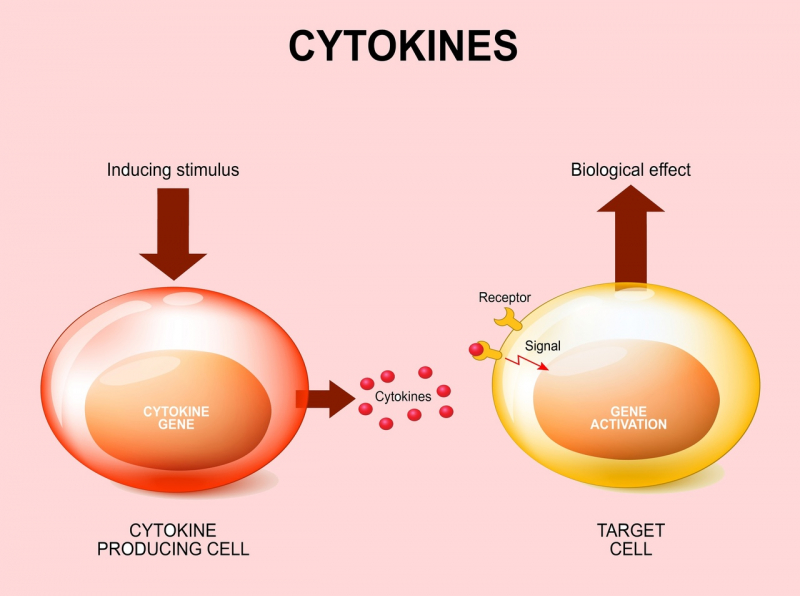
It is a release of pro-inflammatory molecules, cytokines, first of all, by cells of our immune system, but also by other non-specific cells that lead to increased activation of the immune response. We actually need this response to fight viruses and bacterial particles but when the cytokine storm is too strong, excessive activation of the immune system can put healthy tissue at risk, which in turn will lead to a systemic inflammation. That is what is known by the term sepsis.
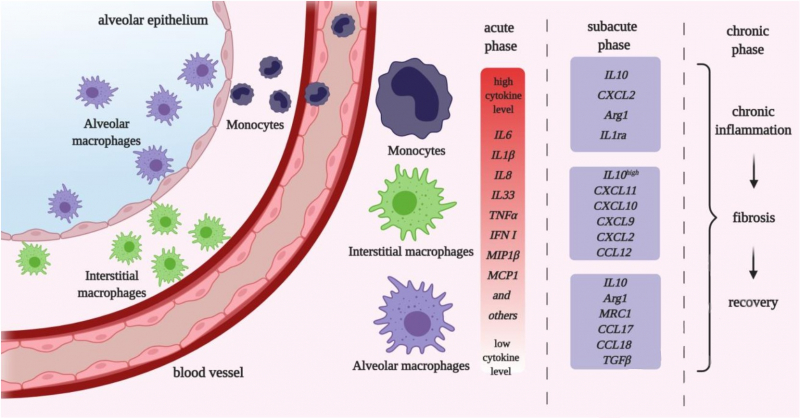
Any strong inflammation causes a cytokine storm, the question is only in its scale. It can be local, it can spread to neighboring organs, or it can take over the whole body: then it means that the cardiovascular system is affected and the blood cytokine level is raised, which can lead to inflammations in other organs (liver, kidneys).
Do researchers know now why viral pneumonias cause such a radical response?
First of all, it has to do with viruses being mutagenic – they can change, and each new version, new stamm, leads to a strong reaction, because the body is not ready to face this new molecule.
Thus, in the body of every patient, there is an uncontrollable spread of virus particles. The more there are, the stronger the immune response – local and systemic.
We are used to thinking that pneumonia during a viral infection is a bacterial complication. That’s why it has to be treated with antibiotics. But, apparently, it’s not entirely true?
In case of an infection, the first to develop is a viral pneumonia. At the same time, the activation of antiviral immunity means that the immune system is weaker when facing other pathogens, which is a risk factor to subsequently catch a bacterial infection.
Moreover, in some cases, if a person has chronic immune diseases, a fungal infection can join the picture. All of this can lead to other pathologies, such as, for instance, intestinal infections.

How pneumonia appears
Your article analyzes mechanisms of immune response. Tell us more about them.
Scientists currently agree on it being a complex process with several populations of cells that take part in it. In our review, we mostly cover macrophages.
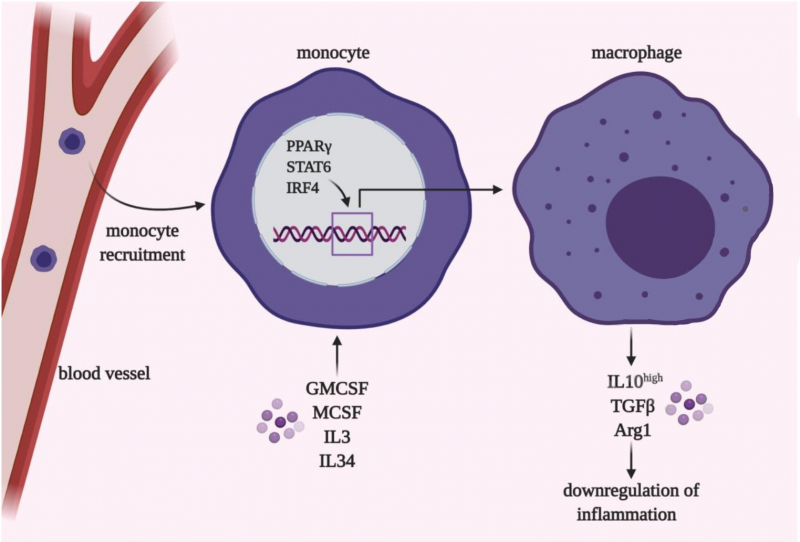
These are cells of innate immunity that are present in all body tissues, including lungs. There are also monocytes or circulating populations in the blood. Monocytes and macrophages are the cells that first interact with a pathogen when it enters our system.
Macrophages and monocytes start producing anti-inflammatory cytokines, the very same ones that form the cytokine storm. Their production is necessary, first of all, to support the immune response against a virus or a bacteria, as well as to form specific adaptive immunity. However, as said above, a cytokine storm, if excessive, is also a risk to other body tissues.
We already know that cytokines are not only produced by cells of the immune system, but also by respiratory epithelium and connective tissues. Moreover, macrophages and monocytes are heterogeneous: there are several subpopulations that have different predispositions to release pro- or anti-inflammatory cytokines.
To put it simply, some populations release a lot of pro-inflammatory mediators and thus are needed to produce the cytokine storm, while others can stop inflammation and take part in regeneration on later stages of pneumonia.
Consequently, the balance between different types of monocytes and macrophages is one of several important factors that determine the outcome of the illness and its seriousness for each patient.
If our bodies have these different types of cells, how does it happen that we develop serious inflammatory processes? Does this balance get skewed?
First of all, there is a certain genetic predisposition. Our review was made in collaboration with Alexey Komissarov, an expert in genomics and head of ITMO’s Applied Genomics lab. The analysis made by Alexey and his colleagues shows that there are certain genetic variations, for instance, in cytokines and their receptors, that are linked to an immune system imbalance, with a higher level of production and activity of pro-inflammatory cytokines.
Second, all populations of monocytes and macrophages are rather flexible. It’s impossible to tell whether a given cell would be pro- or anti-inflammatory. That’s why each patient can have varying numbers of cells with different functions. And this can affect the immune systems’ ability to, first, fight viruses, and second, stop inflammation fast enough, which will determine the seriousness of the illness.
Moreover, by selectively manipulating various immune cell populations, we can successfully both limit the spread of viruses and stimulate regeneration of lung tissue on later stages.
So, does it mean that one person can be genetically predisposed to pneumonia or abscess, while another is not?
We can say that it’s true for some people, while for others there is no proven connection between their genes and the flow of the illness. We cannot say for certain – this connection may yet be found or maybe there are simply other reasons for it.
Do we currently know why macrophages develop their pro- or anti-inflammatory properties? Do we know what can cause the imbalance and an overflow of pro-inflammatory macrophages?
First of all, as I’ve said above, the proportion of pro- and anti-inflammatory populations may be connected to genetic mechanisms.
Second, the level of macrophages activation is connected to the number of virus particles. The more there are and the faster they reproduce, the higher the possibility of a strong inflammatory response.
Finally and most interestingly, virus particles themselves can actively interact with the immune system. They also affect the work of macrophages. Here it all depends on the stamm we are working with.
Does it mean that there are viruses that themselves “turn off” our inflammatory response, while others can stop the release of anti-inflammatory cytokines?
Exactly.

What researchers suggest
As far as I understood, the balance between the two types of cytokines can also be changed with therapy?
Yes, that’s right.
How?
These types of therapy are now all experimental, we cannot regulate the cell populations we target with enough precision. First of all, because the expression of proteins that we target is insufficiently specific. Ideally, we’d want to regulate not a population, but a specific cell. Currently, science and medicine can analyze single cells but not specifically treat it – this very strategy will be key for healthcare of the future.
The most promising methods target transcription factors, i.e. proteins that can connect to DNA and regulate the expression of many genes, such as, for instance, pro- or anti-inflammatory.
We know that there are immunostimulating macrophages and there are others that suppress the immune response. That’s why activating inflammatory activity of specific cells on early stages of the illness for subsequent destruction of the virus seems the most advantageous.
Then, this therapy has to be followed by another one – that facilitates the anti-inflammatory macrophages and lung tissue regeneration, when the virus is already defeated.

And how can we control the functions of macrophages?
We can actually use various approaches. First of all, there are antibodies, chemical inhibitors or activators of specific receptors, signal pathways, or transcription factors in our immune cells. Second, it can be gene therapy, potential editing of DNA (genome) or RNA (transcriptome) of immune cells, but this approach is more applicable for patients with innate immunity problems.
What stage are these methods currently on?
It depends on the situation. The majority of them are currently tested on animals, where many approaches demonstrate their efficiency.
Does it mean we are not to expect such methods to appear in one or two years?
You definitely shouldn’t wait for gene editing of viruses. Chemotherapeutic methods, on the other hand, in the current conditions will most probably be introduced already in 2021.
How much of these efforts are connected with the pandemic?
Naturally, the coronavirus pandemic was a strong impetus for the development of immunotherapy but this research is based on a great amount of previously acquired data.
SARS-CoV-2 is not the first virus that we’ve had to face. Previously, we also had SARS-CoV and MERS epidemics, as well as the annual flu, and they all attract researchers’ attention to viral pneumonia. Their mechanisms are partially similar, which allows us to conduct brilliant translational studies and apply previously developed methods.

Are there any studies showing that viral pneumonia in COVID-19 is different from those caused by other infections?
The basic principles of an antiviral response are rather similar, even though there are differences. These differences are not significant from the point of view of virology and immunology, but they are in terms of therapy – and that is why researchers look at them so closely.
For instance, every virus interacts with the immune system differently. Thus, SARS-CoV-2 suppresses macrophages activation and can successfully avoid immune response until a certain point when the number of particles becomes too big.
Accumulation of many particles leads to a strong cytokine storm. Here it is important to understand that these mechanisms characterise the pathogenesis of COVID-19, but they are not unique for the virus.
What kind of related research is conducted at ITMO?
Our lab studies the regulation of transcription response in monocytes and macrophages in the context of viral infection. We study how cells develop their pro- or anti-inflammatory scenarios. First of all, it has to do with studying the immune response to seasonal pneumonia. We are developing approaches that can help hinder cytokine storms. With this research we are hoping to create our own immune system regulation approach in viral infections