Contents:
- What are carbon dots?
- How were they discovered?
- How do they work and why do they produce light?
- Why are carbon dots an interesting research object?
- Do they have any medical applications?
- How else can they be used?
- How are they synthesized?
- What studies are conducted in the field
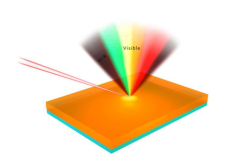
What are carbon dots?

Their one defining feature is their ability to absorb and then radiate light, luminescing in the range from blue to red and even in the infrared spectrum.
How were they discovered?

How do they work and why do they produce light?

Why are carbon dots an interesting research object?

Moreover, the properties of nanoparticles (their size, solubility, the brightness and color of their radiation, etc.) are easy to control not only through manipulating precursors during their synthesis, but also by changing their excitation parameters, such as wavelength.
Thanks to their organic origin, carbon dots are completely eco-friendly, unlike, for instance, perovskite nanocrystals or quantum dots, which contain ions of heavy metals.
Most importantly, however, carbon dots are water-soluble, biocompatible, and have low toxicity.
Do they have any medical applications?

For instance, researchers from ITMO have recently synthesized chiral carbon dots (in other words, incompatible with their mirror copies), which is an important milestone because chirality is an intrinsic property of various biological structures, including DNA, RNA, and protein amino acids. This means that chiral carbon dots may be less likely to be rejected by the human body, while at the same time acquiring the capacity to selectively interact with biological objects. Thanks to these properties, they can be used in diagnostics, including that of oncological diseases, as well as to identify DNA defects and deliver treatment to affected cells.
Another research group from ITMO succeeded in creating nanodots that can absorb and radiate light within the infrared range, which is highly useful given that human tissues are only transparent to waves in this range (800-1,000 nanometers). Such dots can help produce high-quality, detailed images of biological tissues, for example, tumors, inside the body.
How else can they be used?

Another important area of application is using the optical radiation of these particles in optoelectronics, where they can be applied in LEDs on par with semiconductors thanks to their photostability and brightness.
How are they synthesized?

What studies are conducted in the field

In another study, the researchers showed that optical centers of bright red radiation can form on the surface of nanoparticles. Carbon dots acquired by heating citric acid in formamide were observed to possess both green and red luminescence, with optical centers forming both inside nanoparticles and on their surface.