Carbon material
A number of carbon-based materials are well-known to us today. That includes graphite, diamond, fullerenes, carbon nanotubes, and graphene – one of the most resilient yet flexible organic materials. In 2018, the UK-based startup Vollebak unveiled a two-sided nylon jacket with graphene coating. The high-tech outerwear preserves body heat, prevents the growth of bacteria, is windproof, and lowers humidity levels. This and other examples speak to the potential of future research on carbon compounds.
In the mid-‘00s, scientists discovered a new form of carbon called carbon dots. The researchers were drawn in by this material’s bright and persistent emissions. As of today, they have been able to produce emission in the blue-to-green spectrum, but difficulties still present themselves in producing carbon dots of other colors.
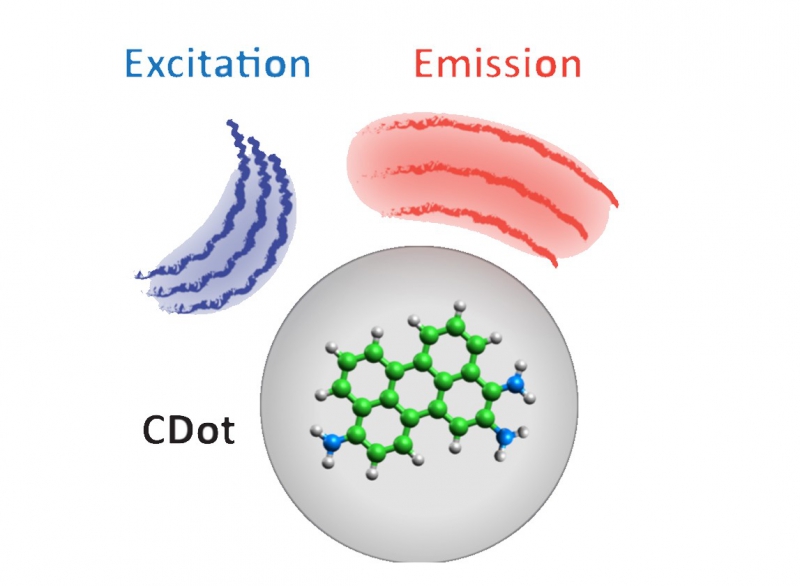
Fluorescent carbon nanostructures
In 2018, ITMO University’s International Research and Education Center for Physics of Nanostructures and Professor Andrey Rogach of the City University of Hong Kong were awarded a megagrant by the Russian Government. The megagrant funding allowed them to found the Laboratory of Light-Emitting Carbon Quantum Nanostructures and conduct the first full-cycle research at ITMO University.
ITMO University’s second-year Master’s student Nikita Tepliakov and his colleague, Evgeny Kundelev, PhD, have published two research papers on carbon dot studies in highly-rated international scientific journals.
“The primary goal of my research was to establish a theoretical model that would describe the key experimental facts relating to carbon dots. We were going off of our understanding of the hybridization of carbon atoms in materials. We know that carbon atoms in diamonds exist in the state of sp3 hybridization, meaning that each carbon atom is linked to four nearby atoms. In graphite, each atom in the state of sp2-hybridization is surrounded by three atoms in-plane and has weak vertical connections. Our study is the first to suggest fractional hybridization. That means that atoms in carbon dots are suspended in a state of intermediate hybridization between sp2 and sp3,” explains Nikita Tepliakov, author of the article published in ACS Nano.

Comparison to experimental data has shown that the researchers’ intermediate hybridization model provides a suitable explanation for the optical properties of carbon dots.
In his article in the Journal of Physical Chemistry Letters, Evgeny Kundelev describes a method of amplifying the red photoluminescence of carbon dots by way of eliminating a fundamental obstacle that prevents its application.
“In my research, I propose a method of synthesizing carbon dots that exhibit strong red photoluminescence. This is achieved by functionalizing their surface with nitrogen in the form of amino groups. That allows us to preserve the carbon dots’ inherent luminescence properties by shifting their emission into the red part of the spectrum,” says Evgeny Kundelev.
Potential applications for carbon dots
Carbon dots present a number of advantages over their counterparts. Firstly, they are quick and cheap to synthesize. For instance, researchers from ITMO University used regular citric acid to synthesize theirs. Secondly, fluorescent carbon compounds make for excellent luminescent marks. Due to their biocompatibility with living organisms, their use presents no harm. Evgeny Kundelev’s research is especially relevant in this regard, as many live tissue cells’ transparency window is located precisely in the red part of the spectrum. What this means is that they absorb blue light, but pass red light through, and that is why only red emission carbon dots can be used as marks.
Fluorescent carbon dots may also be used in sensing of various biological molecules, such as when identifying contaminants in air or water. According to the researchers, carbon dots also have a bright future in the field of optoelectronics. Due to their photostability and bright luminescence, they may be employed in light diodes on par with semiconductors.
Of course, carbon dots also have their applications in medicine. Scientists plan to use them in the targeted delivery of medical products. Their plans involve attachment of special cancer cell-destroying biomolecules to carbon dots which, in their turn, will target tumorous growths. Then, by illuminating them with light, scientists plan to force biomolecules to detach from carbon dots and attack cancer cells.
Researchers at ITMO plan to continue their research on carbon dots by using the models they’ve developed. For one, they will study the effect of various admixtures or functional groups on the optical properties of carbon compounds with the goal of cultivating the desirable optical response.





