Your drug has been attracting a lot of interest lately, with its information video gaining over 800,000 views on YouTube. Can you, first of all, tell us more about blood clots, their danger and why they attract so much attention?
Head of the Regional Vascular Center, operating surgeon at the Mariinskaya Hospital Ivan Dudanov: The urgency of the problem is such that it cannot be overestimated. If we analyze the causes of all, as we say, adverse outcomes, that is, fatalities, serious complications, disablements, then thrombosis and embolism are the most common. They account for about two-thirds of all adverse outcomes.
Thrombosis is the formation of a dense clot on the wall of a vessel. If it occurs in an artery that brings blood to a particular organ, this causes a decrease in blood supply to that organ, and this is how local asphyxia develops. If the vessel’s opening closes completely, this leads to the death of the organ or part of the organ. For example, a stroke is caused by the formation of a dead part in the brain, while a heart attack by the formation of a dead part in the myocardium. If a blood clot is small, it can break loose and clog smaller vessels. The result is the same – the death of an organ or its part. If a blood clot forms in a vein, it complicates the blood outflow process, which causes complications in the form of edema, the formation of post-thrombotic disease, these are very severe disabling pathologies.
The factors that lead to the formation of blood clots can be very different: oncologic pathologies, other illnesses, even dehydration. This can also be caused by the uptake of a range of medical drugs.
What is the current treatment of thromboses, and why did you decide to develop a new drug?
Head of ITMO University’s SCAMT Center Vladimir Vinogradov: The methods of dissolving blood clots by means of special thrombolytic drugs have been in place since the end of the 20th century. Upon entering the body, they cause a cascade of reactions, which leads to the formation of plasminogen which is contained in our blood, plasmin. It is this proenzyme that, in fact, dissolves a blood clot.
However, as a chemist, I can say that their use presents us with two important problems. The first is that such drugs have a very short lifetime in the human body: as a rule, they can remain active for no more than five to ten minutes after their administration. Following that, these molecules are deactivated by the body, which interprets them as a foreign substance which it tries to neutralize as quickly as possible. This effect can only be negated by administering a fairly large amount of the drug so that it can remain active until it begins to operate at the site of thrombosis. But then a second problem arises – if you introduce too much drug, the so-called hemorrhagic complications may appear, in other words, this can cause bleeding – from the gums, eye sockets, nails, internal bleeding. All this results in a situation that if you administer a small dose, it won’t work, if you administer a large one, you’ll cause bleeding.
Ivan Dudanov: Another problem of this method is that it’s only effective when administered in the first two–four hours since the formation of the blood clot, during the so-called therapeutic window. Because of this, the number of patients that undergo such a treatment only amounts to 4–6%. You have to manage to deliver a patient from their home to the hospital, examine them, diagnose them and inject a thrombolytic substance, at this time. That’s why in the majority of cases other methods of blood clot removal are used.

Will your development solve these problems?
Vladimir Vinogradov: At SCAMT, we adhere to the concept that there is no need to create new treatment molecules if there are already ones that work. Plasmagene activators already exist, and they perform their immediate function of dissolving a fibrin clot (thrombus) rather well. The question that remains is how to deliver them safe and sound to the place where this drug is needed.
We came up with a unique composition that combines the thrombolytic effect, the anticoagulant effect, and the possibility to control the movement of molecules in the body with the help of a magnetic field. Anticoagulants are a substance that prevents the formation of new blood clots. Typically, such drugs should be prescribed in combination with thrombolytic treatment. However, one related problem is that anticoagulants also lead to a loss in the activity of thrombolytic enzymes, so combining these two groups is not an easy task.
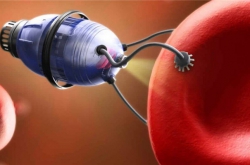
What we created is a drug that can be delivered to the site of a blood clot formation by means of a magnetic field, where it then activates plasminogen and, after that, prevents the formation of new blood clots. We haven’t invented a new pharmaceutical molecule. The uniqueness of our development consists precisely in the composition of these three components.
The process of administering our drug is similar to the one currently used: a vein injection. But after this, the substance moves under the influence of a magnetic field along a directed trajectory to the place where the blood clot is detected.
How is this composite created?
Vladimir Vinogradov: We have three main components. The first is the magnetite nanoparticles. This substance has been approved for introduction into the human body, it’s used as a contrasting reagent during MRI. These particles provide magnetic control. The second component is a plasminogen activator of the urokinase type, that is, the active substance that contributes to the dissolution of the clot. Last but not least is heparin, which prevents the formation of new blood clots.
We’ve found a scheme that allows to stitch these three components together in an optimal way. The main operating load here is performed by the magnetite: it’s important not to overload it with an organic component so that it doesn’t lose the ability for effective control via a magnetic field.
In 2018, the news appeared that the drug had undergone preclinical tests – what were the results and how were they conducted?
Vladimir Vinogradov: As of now, the drug has indeed passed the stage of having been tested on cells and artificial blood clots. We conducted full preclinical trials. These included evaluations of pharmacokinetics, pharmacodynamics, efficacy, mutagenicity and a range of other aspects. The drug was administered to animals; in all these cases, tests on already existing drugs were conducted to ensure control.
As a result, indicators of toxicity and side effects of the drug turned out to be lower than those of the ones currently used. This is due to the fact that by delivering the drug directly to the site blood clot formation, we can get away with a much smaller amount of the substance.

Ivan Dudanov: During these tests, it’s necessary to evaluate not only the quality but also the speed of the action. We observed in prostheses sewn into animals that when using our drug no traces of thrombosis were left at all, even though the existence of the blood clot in the prostheses was measured not by minutes, hours and days but significantly longer periods. At the same time, the animals continued to live a full life since the injected dose of the drug was atoxic. During the tests, it turned out that this drug should be in the amount that is 100 times lower than that of its existing counterparts, with its simultaneously being 4000–4500 times more effective.
Is it true that some of the tests were done at the Mariinskaya Hospital?
Ivan Dudanov: Yes, some research was done there. Of course, we’re not talking about using the drug on actual patients. These tests were done using expendable materials in the form of coagulated blood clots in test tubes. These are test tube remnants of blood samples that are taken from patients as part of biochemical and clinical tests and then disposed of at the end of the study.
It’s these clots that we used, they were depersonalized, there were no patients data on them. For us, the clots were what’s important – their sizes, their lifetime, etc. Such tests are significant because you can use animal blood in the early stages, but for the tests after that human blood is required.
What work needs to be done next before the drug goes to market?
Vladimir Vinogradov: The question that stands now is how we are to go into clinical trials. In preclinical trials, we can use a regular magnet, but in the clinic, we have to use only the equipment that has been authorized by the Ministry of Health, whether it is a scalpel, a tomograph or a magnetic installation.
At the same time, I’m aware of only one magnetic device that meets our criteria. It’s now being created in Germany and undergoing the certification process Europe-wide. It’s not yet available in Russia. All this requires significant investments. There’s also a number of other problems related to the fact that we’re not creating a new molecule but using an existing one.
Ivan Dudanov: We still have a huge number of tasks to do – there is much to learn in the course of the impending clinical trials. Supposing that we have administered the drug, there is a magnetic field and the drug is circulating in the body, we need to establish when the drug will be removed from the body and what effect it will have on the internal organs. There’s another question: following its effective dissolution of the blood clot, would there be a fragmentation effect when the blood clot falls apart into small pieces that clog other vessels? Without answers to these questions, we simply can’t carry out, let alone expand on, our research.
There could also be contraindications – diseases or other specific conditions that wouldn’t allow for the drug to be used. All this requires careful consideration. That’s why we can’t yet say that we’ve invented a panacea that could be used starting from tomorrow.

When are you planning to start the clinical trials and how much time will they take?
Ivan Dudanov: This is a very tricky question as there are various legal aspects to consider. It’s also necessary to calculate all the possible scenarios of how the drug would behave in the human body.
On top of that, it should also be taken into account that the clinical trials will definitely involve volunteers. We’ll have to obtain the patients’ consent to participate in the experiment, create groups of patients differentiated by the type and severity of pathologies and so on. Also integral are long-term observation of patients over time and studying the drug’s long-term results.
Vladimir Vinogradov: It’s not only us that the timing depends on: on our part, we’re ready to start and provide everything we can for the purposes of the trials. But as my colleague has already mentioned, the key question is legal nuances. It’s also necessary to understand the specifics of the global pharmaceutical market, which is that the development of a drug is always a very risky business. If a pharmaceutical company feels that the project is very risky, it’s extremely difficult to attract it on board as an investor. We’re looking for partners, we’re considering a public-private partnership, we’re ready to search for those who can develop the necessary magnetic equipment.
Now we’re in the midst of negotiating, and most of our negotiations are with European companies. But we can’t state with absolute certainty that tomorrow they’ll come and provide us with investments. We are in an active search for an investor who would help us implement both clinical research and the development of the equipment. The clinical trials will take at least five years.